The so-called smart Kd‑concept treats radionuclide sorption processes in natural systems, whereby the smart Kd‑values are mainly based on mechanistic surface complexation models (SCM) and ion exchange (IEx). The distribution coefficients may thus vary in time and space depending on the actual geochemical conditions. As this parameter space is indeed huge for all real-world application the challenge of identifying (and parameterizing) the most relevant ones must be solved. Sorption is described by the “component additivity approach” (bottom-up strategy), in which the contribution of the sorption of one element is considered on every single mineral fraction of a sediment, also including relevant competitive effects.
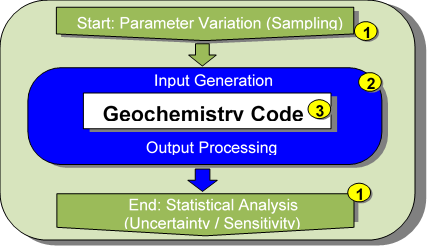
The computational scheme producing smart Kd‑values is as follows: An outer shell varies all parameters considered important within their boundaries as imposed by field experiments. Each representation of the combined input parameter vector is then passed on to a geochemical speciation code (often utilizing a suitable ASCII data processing tool as intermediate layer) that computes the smart Kd‑value associated to this very parameter vector. This is then repeated for all parameter combinations, see Fig. 1 for a generic scheme, naming also the codes currently used for the application cases. The fundamental strategy for the generation and utilization of the smart Kd-matrices is described in detail in Stockmann et al. (2017).
Fig. 1: Coupling procedure to calculate multidimensional lookup tables for smart Kd-values, with SIMLAB as 1, UCODE as 2 (Poeter et al., 2014) and PhreeqC as 3 (Parkhurst and Appelo, 2013) in the current implementation.
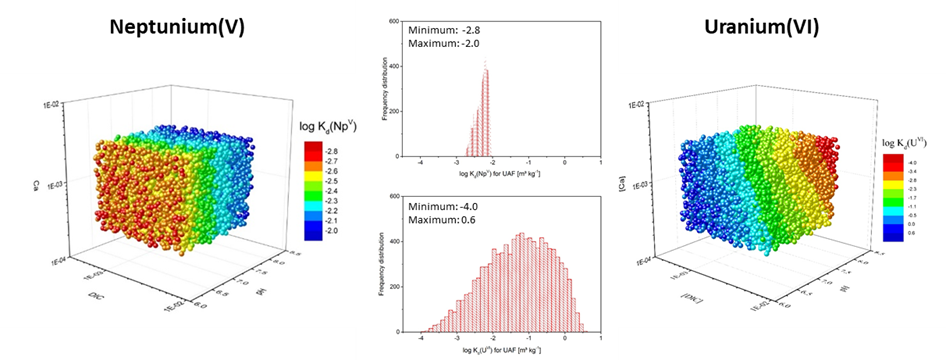
This highly automated process behind the computation of smart Kd‑values easily allows to set-up extensive uncertainty and sensitivity analysis. As an illustrative example for uranium and neptunium sorption onto a sediment of the Gorleben cap rock (see Applications) is shown in Fig. 2. The dependence of the smart Kd-values on pH, dissolved inorganic carbon (DIC) and Ca-concentration is evaluated (realized with random sampling generating 10,000 parameter sets). For U, the DIC and pH mainly influences the sorption. The Kd‑values decrease with increasing pH due to the sorption maximum in the near neutral pH range. Furthermore, combined effects are visible depending on the Ca content and the DIC of the groundwater type. For Np, the pH is the most sensitive parameter concerning the sorption. The Kd will be higher with increasing pH up to the sorption maximum between pH 8-9 before it lowers in the more alkaline conditions. From the histograms it is obvious that the log Kd for U range over several orders of magnitudes compared to the smaller range for Np, which is related to the complex chemistry of uranium.
Fig. 2: Multidimensional Kd-matrix for Np(V) (left) and U(VI) (right) sorption in the Gorleben upper aquifer sediment as a function of pH, dissolved inorganic carbon (DIC), and Ca-concentration (Kd in m³ kg-1, logarithmic scale). The middle column gives the histograms of all 10,000 computed smart Kd‑values useful for uncertainty analysis.
References:
Parkhurst, D. L., Appelo, C.: Description of input and examples for PHREEQC version 3: A computer program for speciation, batch-reaction, one-dimensional transport, and inverse geochemical calculations. In U.S. Geological Survey Techniques and Methods, book 6, volume A43 of Modeling Techniques. U.S. Geological Survey, Denver, Colorado, 2013.
Poeter, E. P., Hill, M. C., Lu, D., Tiedeman, C. R., Mehl, S.: UCODE_2014, with New Capabilities to Define Parameters Unique to Predictions, Calculate Weights using Simulated Values, Estimate Parameters with SVD, Evaluate Uncertainty with MCMC, and More (2014).
Stockmann, M., Schikora, J., Becker, D.-A., Flügge, J., Noseck, U., Brendler, V. (2017): Smart Kd-values, their uncertainties and sensitivities - Applying a new approach for realistic distribution coefficients in geochemical modeling of complex systems. Chemosphere 187, 277-285.