Sorption characteristics of Ni(II) were investigated under varying geochemical conditions. Therefore batch sorption experiments (http://www.smartkd-concept.de/?q=Experimental%20program) onto quartz, orthoclase and muscovite were conducted. Collected sorption data will be used for determination of surface complexation constants by inverse modeling with the geochemical speciation code PHREEQC in combination with the parameter estimation code UCODE.
The following geochemical parameters were varied (Tab. 1):
- the initial concentration of Ni (C0)
- th pH
- the solid-to-liquid ratio (M/V)
- the ionic strength and
- the background electrolyte (sodium perchlorate used as an inert background solution, sodium sulphate used to include ligand influence on Ni sorption).
Tab. 1: Matrix of batch experiments to investigate Ni sorption onto quartz, orthoclase, muscovite.
As an example, results of quartz batch experiments are illustrated in Fig. 1. Generally, results show expected sorption tendencies with respect to sorbed fractions of Ni and pH, C0, M/V and ionic strength: The higher the pH and the lower the initial Ni concentration, the more sorption occurs. Furthermore, a higher M/V results in higher sorbed Ni fractions. Moreover a higher ionic strength results in lower sorbed fractions. Results reveal a weak sorption preference of Ni towards quartz surfaces; the interrelation between ionic strength and Ni sorption might indicate the formation of outer-sphere complexes which has to be confirmed with spectroscopic measurements in detail.
As shown in Fig. 1 B, batch experiments using quartz and Ni in 0.01 and 0.2 M NaClO4 background solution resulted in negative sorption values at pH 5 and 6. This could be caused by a release of Ni from the quartz sediment due to dissolution processes (quartz dissolution data not shown here).
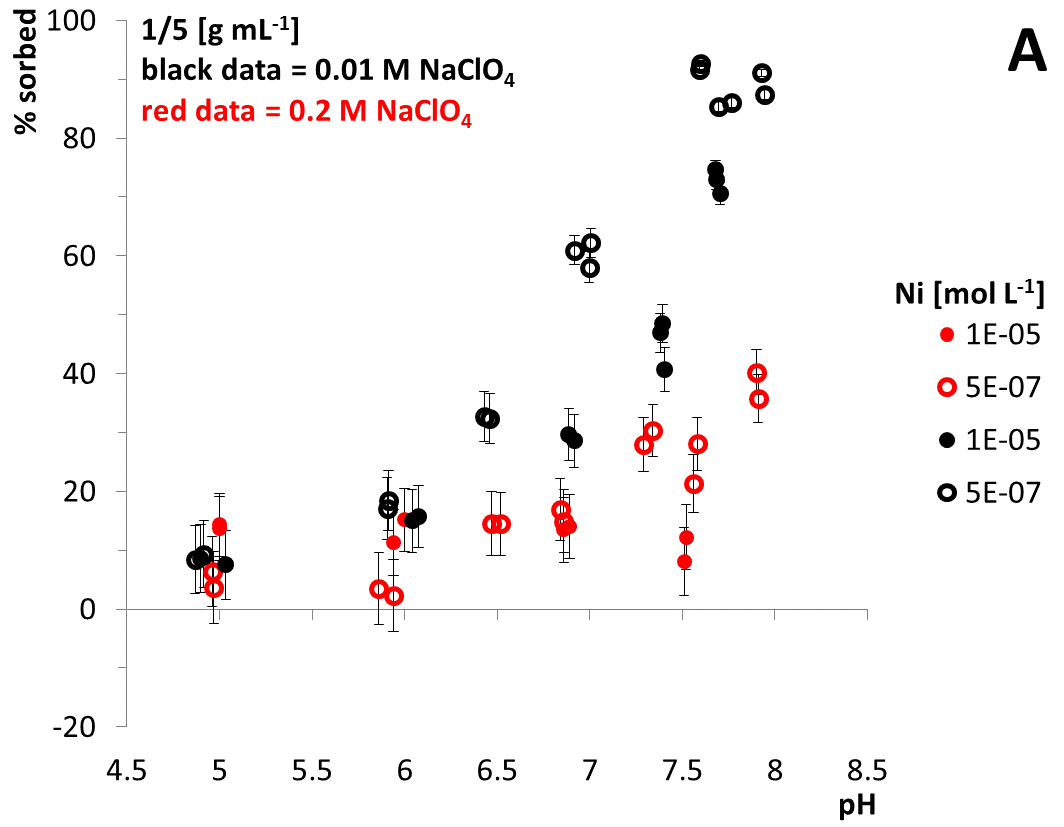

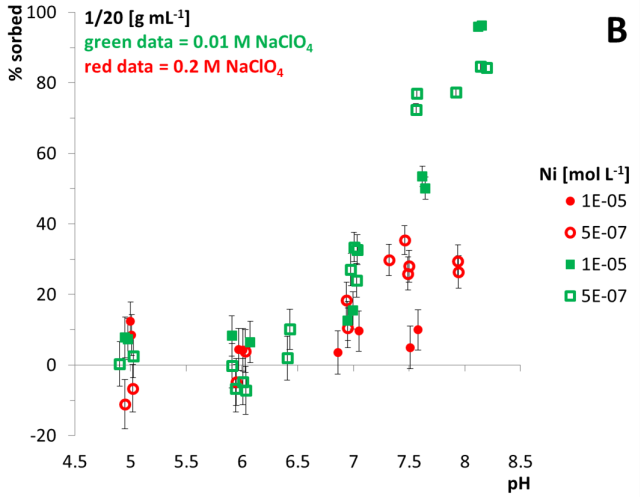
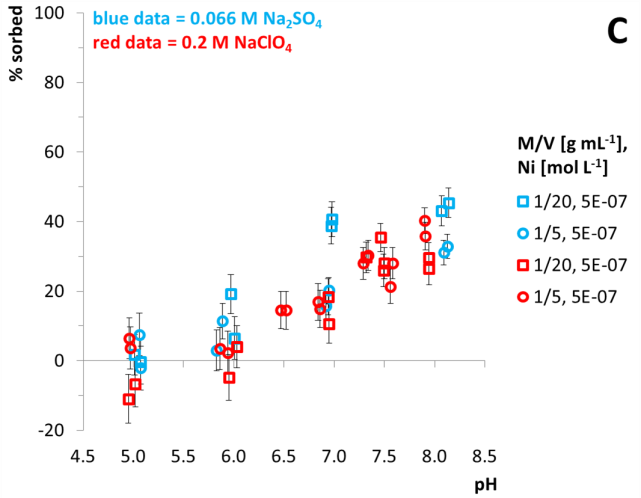
Fig. 1: Results of batch experiments applying quartz and Ni(II). A: M/V 1/5 g mL‑1, B: M/V 1/20 g mL‑1, impact of ionic strength. C: impact of SO42- as a ligand. Twofold measurements are conducted. Equilibration time = 3 d. Vertical error bars are calculated using 10‑7 mol L‑1 Ni long term precision measurements of the ICP-MS.
Additional batch experiments with quartz are conducted to investigate the impact of SO42- as a ligand. Using the same ionic strength compared to 0.2 M NaClO4 a 0.066 M Na2SO4 background solution was applied. Sulphate does not seem to effect Ni surface complexation (Fig. 1, C). This was not expected since speciation calculations reveal NiSO4 as the main aquatic species with nearly 56 % abundance in the pH range 5 ‑ 8 (Fig. 2, A). In 0.2 M NaClO4 the main species is Ni2+ (Fig. 2, B). Above pH 6 NiSiO(OH)3+ is also an important aquatic phase since 4∙10-5 mol L-1 Si was determined to be lowest measured Si concentration in all batch systems (data not shown here). Evaluation of these batch experiments is still in progress.
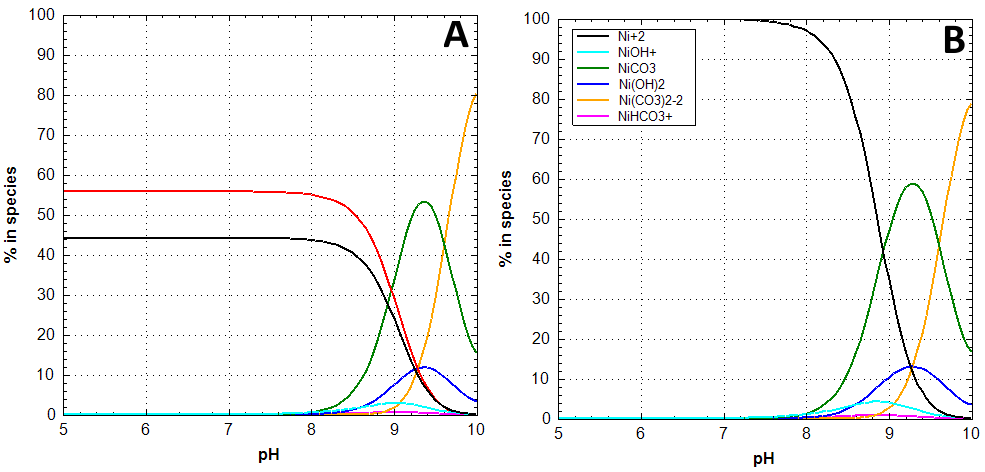
Fig. 2: Ni speciation diagrams with and without SO42- in solution. A: 0.066 M Na2SO4, B: 0.2 M NaClO4. Atmospheric conditions, 10‑5 mol L‑1 Ni.
Back to Batch Experiments